Reaction
Chemical Modification of Nucleotides
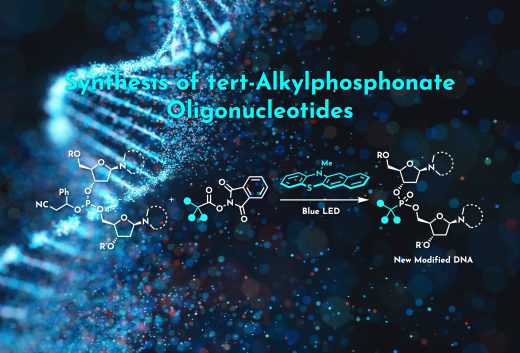
Chemical modification of nucleotides can improve the metabolic stability and target specificity of oligonucleotide therapeutics, and alkylphosphonates have been employed as charge-neutral replacements for naturally-occurring phosphodiester backbones in these compounds. However, at present, the alkyl moieties that can be attached to phosphorus atoms in these compounds are limited to methyl groups or primary/secondary alkyls, and such alkylphosphonate moieties can degrade during oligonucleotide synthesis. The present work demonstrates the tertiary alkylation of the phosphorus atoms of phosphites bearing two 2’-deoxynuclosides. This process utilizes a carbocation generated via a light-driven radical-polar crossover mechanism. This protocol provides tertiary alkylphosphonate structures that are difficult to synthesize using existing methods. The conversion of these species to oligonucleotides having charge-neutral alkylphosphonate linkages through a phosphoramidite-based approach was also confirmed in this study.