Published to Precision Chemistry
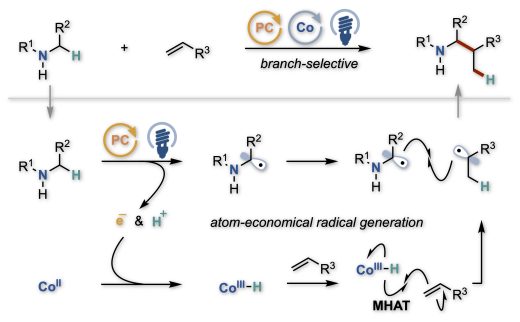
We report a visible-light-driven dual photoredox/cobalt catalytic system that enables Markovnikov-selective hydroaminoalkylation of alkenes. This cooperative catalysis generates and couples two radical species derived from alkylamines and alkenes via sequential single-electron and proton transfers (SET/PT) from α-amino C(sp³)–H bonds of amines to alkenes. The method enables the installation of secondary or tertiary alkyl groups at a-amino positions of a-amino acid derivatives, peptides, and benzylaniline derivatives using a broad range of alkene partners. This strategy represents a conceptually distinct, redox-neutral, and atom-economical approach to hydrocarbofunctionalization beyond conventional C–H activation pathways.
DOI: 10.1021/prechem.5c0038